Bevacizumab
Other name
BTD00087, BIOD00087
International/Other brands
Avastin
Groups
Approved
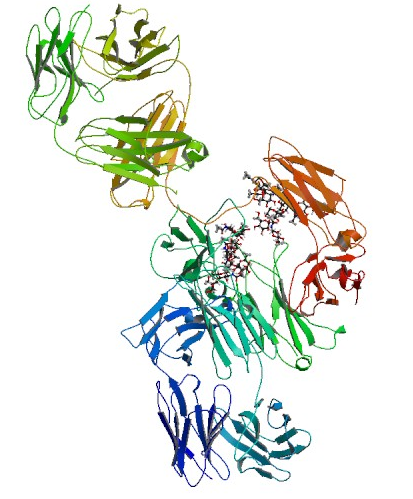
Structure
Prescription products
| Name | Dosage | Strength | Route | Labeller |
|---|---|---|---|---|
| Avastin | Solution | 25 mg | Intravenous | Hoffmann La Roche |
| Avastin | Injection, solution | 400 mg/16mL | Intravenous | Genentech, Inc. |
| Avastin | Injection, solution | 100 mg/4mL | Intravenous | Genentech, Inc. |
Target
-
Description
A recombinant humanized monoclonal IgG1 antibody that binds to and inhibits the biologic activity of human vascular endothelial growth factor (VEGF). Bevacizumab contains human framework regions and the complementarity-determining regions of a murine antibody that binds to VEGF. Bevacizumab is produced in a Chinese Hamster Ovary mammalian cell expression system in a nutrient medium containing the antibiotic gentamicin and has a molecular weight of approximately 149 kilodaltons.
Indications
As part of combination therapy for metastatic colorectal cancer and HER2-negative metastatic breast cancer.
Other indications
-
Mechaism of action
-
Absorption
-
Metabolism
Most likely removed by opsonization via the reticuloendothelial system when bound to endothelial cells, or by human antimurine antibody production
Toxicity
Category C in pregnancy. No studies on lactating patients: recommended to discontinue the drug or discontinue lactation. No extensive studies in pediatrics. In geriatrics: increased risk of proteinuria, arterial thromboembolic events, as well as GI bleeding and sepsis among others.