Afatinib
Other name
-
International/Other brands
Gilotrif
Groups
Approved
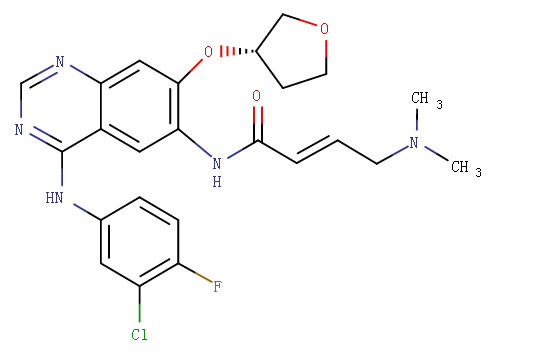
Structure
Prescription products
| Name | Dosage | Strength | Route | Labeller |
|---|---|---|---|---|
| Gilotrif | Tablet, film coated | 20 mg/1 | Oral | Boehringer Ingelheim |
| Gilotrif | Tablet, film coated | 40 mg/1 | Oral | Boehringer Ingelheim |
| Gilotrif | Tablet, film coated | 30 mg/1 | Oral | Boehringer Ingelheim |
| Giotrif | Tablet | 20 mg | Oral | Boehringer Ingelheim (Canada) Ltd Ltee |
| Giotrif | Tablet | 40 mg | Oral | Boehringer Ingelheim (Canada) Ltd Ltee |
| Giotrif | Tablet | 30 mg | Oral | Boehringer Ingelheim (Canada) Ltd Ltee |
Target
Description
Afatinib is a tyrosine kinase inhibitor which is a 4-anilinoquinazoline. It is prepared has the dimaleate salt. FDA approved on July 12, 2013.
Indications
Metastatic Non-Small Cell Lung Cancer; Refractory, metastatic squamous cell Non-small cell lung cancer
Other indications
-
Mechaism of action
-
Absorption
Following oral administration, time to peak plasma concentration (Tmax) is 2 to 5 hours. The geometric mean relative bioavailability of 20 mg tablets was 92% as compared to an oral solution. Food decreases Cmax and AUC relative to the fasted state.
Metabolism
Enzymatic metabolism of afatinib is minimal. Covalent adducts to proteins are the major circulating metabolites.
Toxicity
Most common adverse reactions (≥20%) are diarrhea, rash/dermatitis, acneiform, stomatitis, paronychia, dry skin, decreased appetite, pruritus.